|
Less than the density of pure water, but more than the density of stock 70 alcohol solution o less than the. Write these facts on the board: Pure water has a density of 1g/cm. Question 3 1 pts Based on your data for the solution density of the diluted alcohol solutions (alcohol/water mixtures), the densities of all dilute alcohol solutions should be. 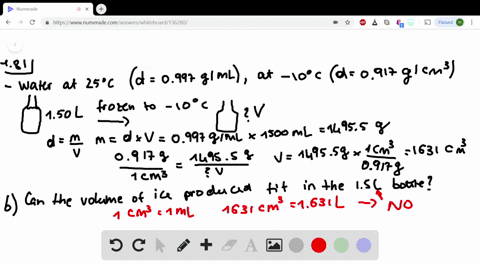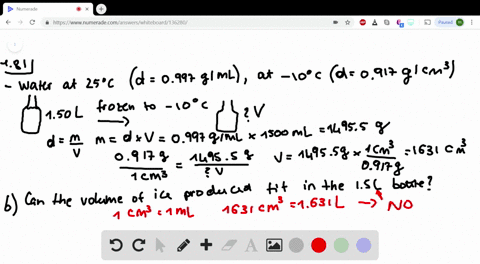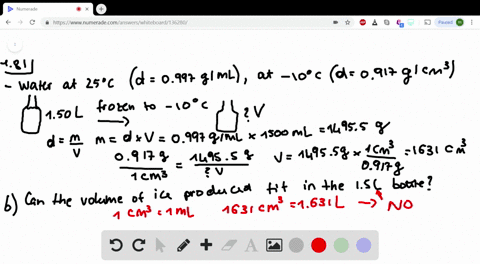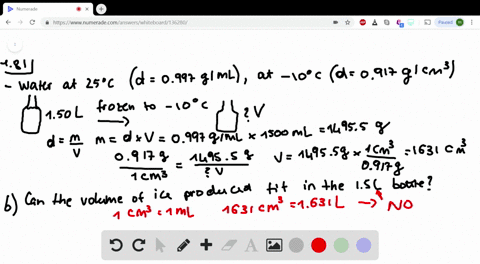 
Trending Questions Is sodium hydroxide and bleach the same thing? What does sulfate look like? Each component in a mixture maintains its own characteristics? When was ACS Chemical Neuroscience created? What mineral composed only of oxygen and silicon? What is the boiling point of ketamine? How many elements does hcl? How many molecules are in 334 g CBr4? Why is picric acid is more acidic than phenol? Is volume conserved in a reaction between distilled water and ethanol? Why does carbon dioxide move from blood to alvelous? What kind of bond is Na with Na? What are elements made out of? Is sodium a nonmetal or a metal or a solid or a liquid? How to calculate the standard state Gibbs Free Energy values at pH 0 and for dissociation of acetic acid with a Ka 1. Saltwater is more dense than isopropyl alcohol, so the alcohol floats on top of the saltwater.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
